Featured Projects

Innovative Telehealth Solutions for Veterans with Chronic Pain Learn More >
A Prospective Investigation of the Risks of Opioid Misuse Learn More >
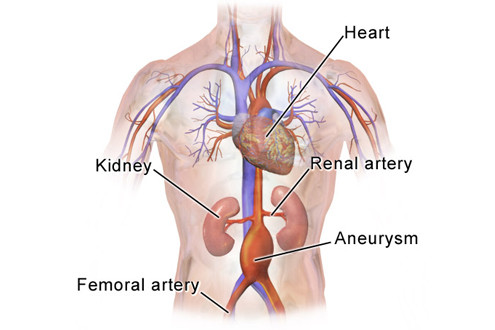
Nicotine and aortic aneurysm, a cross-generational problem? Learn More >
Targeted Strategies to Accelerate Evidence-Based Psychotherapies Implementation in Military Settings Learn More >
Predicting Resilience in the Human Microbiome Learn More >


